Ph.D.
- FMA
- The Fabricator
- FABTECH
- Canadian Metalworking
Categories
- Additive Manufacturing
- Aluminum Welding
- Arc Welding
- Assembly and Joining
- Automation and Robotics
- Bending and Forming
- Consumables
- Cutting and Weld Prep
- Electric Vehicles
- En Español
- Finishing
- Hydroforming
- Laser Cutting
- Laser Welding
- Machining
- Manufacturing Software
- Materials Handling
- Metals/Materials
- Oxyfuel Cutting
- Plasma Cutting
- Power Tools
- Punching and Other Holemaking
- Roll Forming
- Safety
- Sawing
- Shearing
- Shop Management
- Testing and Measuring
- Tube and Pipe Fabrication
- Tube and Pipe Production
- Waterjet Cutting
Industry Directory
Webcasts
Podcasts
FAB 40
Advertise
Subscribe
Account Login
Search
Combating plate corrosion
Improving corrosion resistance through welding, fabrication methods
- By Hira Ahluwalia
- October 9, 2003
- Article
- Metals/Materials
According to a recent study sponsored by the U.S. Federal Highway Administration (FHWA)1, with support from NACE International—The Corrosion Society, corrosion-related direct costs such as prevention methods and infrastructure repair and replacement make up 3.1 percent of the gross domestic product (GDP)—a value of about $276 billion annually (see Figure 1). This comes out to nearly $1,000 per person each year. The study estimates that 25 to 30 percent of the cost is entirely preventable.
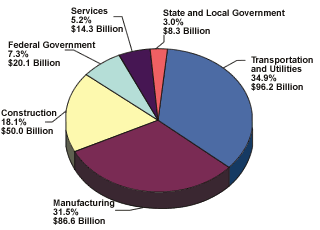 |
| Figure 1 The total direct corrosion costs for each of the Bureau of Economic Analysis (BEA) industry categories is about $276 billion annually. Indirect costs of corrosion are conservatively estimated to be equal to the direct costs, for a combined cost of approximately $551 billion annually, or 6.3 percent of the GDP. |
Unfortunately, damage caused by corrosion is not just a financial burden to society—it has an impact on safety (see Corrosion Accidents at the bottom of this page). For example, in an incident several years ago, the collapse of a 2,000-cubic-meter liquid petroleum gas sphere killed one person and seriously injured another. Investigation of the accident identified that severe corrosion on the sphere's legs had reduced their thickness by up to 8 mm, with pit holes of up to 10 square centimeters.
Corrosion can be broadly defined as deterioration of a material by a chemical or electrochemical attack. Metallic corrosion under aqueous conditions can take place by many mechanisms with a varied impact on the integrity of the material.
Different metals corrode for different reasons, each with its own mechanisms. Understanding the corrosion degradation mode—general, localized, environmentally assisted cracking, intergranular—is key to controlling and preventing corrosion. Careful material selection, good design, and quality fabrication can help prevent the most serious corrosion problems and extend the lifetime of a component in a corrosive environment.
General Corrosion
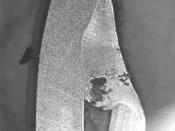 |
| Figure 2 Pitting, crevice attack, and microbiologically influenced corrosion (MIC) all are localized corrosion. |
General corrosion is a uniform loss of material from the surface of a metal and is the most commonly encountered type of corrosion. The metal gradually becomes thinner and eventually loses structural integrity.
The following methods should be considered to minimize general corrosion:
- Choose the best construction materials, and then assign a corrosion allowance to the equipment that is being used. A corrosion allowance is extra thickness added to the wall to compensate for the metal expected to be lost over the equipment's life.
- In many corrosive environments, the weld metal may be preferentially attacked because the cast structure of the weld can be quite different from the parent wrought structure. To avoid preferential corrosion of the weld metal, use filler metals with higher alloying content.
Localized Corrosion
Conditions may exist that cause corrosive attack in a specific location or under a specific set of conditions (see Figure 2). Such attack normally is referred to as localized. Pitting, crevice attack, and microbiologically influenced corrosion (MIC) all are localized corrosion.
Pitting. Pitting is a form of localized attack that results in the formation of holes on the metal surface that often can propagate quickly, leading to material perforation and failure in a short time period. The microenvironment within the pit itself can be autocatalytic, making this a particularly dangerous form of attack. The chloride ion is a particularly aggressive pitting agent for many alloy systems.
Crevice Attack. Crevice attack usually takes place in a tight gap between two surfaces. Similar to pitting, the microenvironment within the crevice can differ greatly from the general environment. Concentration cells can cause this type of corrosion to progress rapidly.
MIC. MIC is responsible for the deterioration of a metal surface through crevice corrosion. Certain types of bacteria form dome-shaped colonies on the metallic surface. The inside of the structure is sealed from the outside. The life cycle of the bacteria produces a corrosive environment within the colony, which causes crevice attack of the metal.
The following methods may minimize localized corrosion:
- Weld shut all crevices.
- Use double-butt or double-lap welded joints.
- Use consumable/removable inserts for critical single-butt joints.
- Don't use skip welding.
- Seal-weld all tubes to tube sheets.
- If crevices cannot be sealed or eliminated, open them up to allow air circulation.
- Remove backing rings in pipe welds after welding.
- Remove embedded iron in stainless steel (SS) equipment.
- Remove surface oxides produced by welding, including heat tints.
- Remove arc strikes, weld spatter, and slag particles.
 |
| Figure 3 The residual stresses from the weld played a role in the chloride stress corrosion cracking in this 304L stainless steel weldment. |
Environmentally Assisted Cracking
Some types of corrosion degradation take place as a result of a synergistic effect between the chemical environment and the mechanical condition of the metal itself. The common forms of environmentally assisted cracking are stress corrosion cracking (SCC) and corrosion fatigue.
SCC. Stress corrosion cracking is the brittle fracture of a susceptible material under tensile stress in a specific environment over a period of time. During this type of cracking, most of the metal surface is virtually unattacked, while fine cracks propagate through the cross section. Chloride SCC of SS is an example of this type of attack (see Figures 3 and 4).
SCC is insidious because usually no trouble manifests before cracking appears. Failures of this type can be unexpected and hence hazardous and expensive. It is a major problem in the chemical and petrochemical industries.
Corrosion Fatigue. Corrosion fatigue is defined as a combination of normal fatigue and corrosion that causes failure below the metal's normal endurance limit. Thus, it is normally encountered not as a visible degradation of the metal but as a premature failure of a component under cyclic conditions.
 |
| Figure 4 A photomicrograph shows the transgranular nature of chloride stress corrosion cracking. |
To minimize SCC and corrosion fatigue, consider the following methods:
- Stress-relieve susceptible material after welding. This heat-treatment procedure will lower the level of residual tensile and cyclic stresses in the component.
- Create compressive stresses by shot peening, which puts the outer surface in compression. Because fatigue and SCC require tension stresses, greater resistance can be obtained by shot peening.
- Avoid stress raisers by blending welds to the level of the base metal.
- Use butt welds instead of fillet welds, because fillet welds are much more susceptible to corrosion fatigue and SCC.
- Blend grinding marks, rough edges, and rough machine marks.
- Avoid unnecessary cold work. Cold-working the material using various fabrication methods increases the residual stresses that may make a component more susceptible to corrosion fatigue and SCC.
Intergranular Corrosion
Intergranular corrosion is the selective attack of a metallic component at the grain boundaries by a corrosive medium (see Figure 5). Several conditions can cause a material to be susceptible to intergranular corrosion. Thermal mechanical processing may cause metallic compounds to precipitate and migrate to grain boundaries. If these are more reactive than the metallic matrix, they can be selectively attacked.
 |
| Figure 5 Intergranular corrosion of nickel 200 in the vicinity of a weld was caused by graphitization of the grain boundaries. |
A commonly encountered form of intergranular corrosion is the attack of nonstabilized austenitic SS caused by the formation of chromium carbide (Cr23C6) precipitates and the subsequent depletion of chromium. Nickel 200 (UNS N02200) is also prone to intergranular attack when exposed to high temperatures in certain environments. In this case, graphitization of the grain boundary leads to the intergranular attack.
These methods may help minimize intergranular corrosion:
- Use stabilized or low-carbon welding materials.
- Do not stress-relieve unstabilized SS in the carbide precipitation range—specify an annealing treatment instead. Stress-relieving temperatures can readily lead to the formation of chromium carbides in the grain boundaries.
- Specify low-carbon or stabilized grades of SS.
Welding Corrosion-resistant Alloys
Although many of corrosion-resistant alloy groups exist, this section briefly discusses welding of austenitic SS, duplex SS, and nickel alloys.
Austenitic SS, such as the 300 series, has been the workhorse of many industry sectors. The corrosion resistance of these alloys is quite good unless the media contains halide ions. Halide ion, particularly chloride, makes the alloys susceptible to chloride SCC and localized corrosion.
Two major goals when welding austenitic SS are to make the welds crack-free and to ensure that the weld and heat-affected zone (HAZ) have the same corrosion resistance properties as the base metal. Selecting the correct filler material, controlling the heat input and weld bead contour, and ensuring the presence of small amounts of delta ferrite in the weld microstructure significantly improve the corrosion resistance of weldments.
Welding procedures and heat treatments must be taken into consideration to obtain sound, corrosion-resistant welds.
In corrosive environments in which austenitic SS is not corrosion-resistant and is susceptible to SCC, duplex alloys such as UNS S31803 and S32205 are increasingly being specified. Duplex SS is an alloy family that has two phases—ferrite and austenite—with ferrite typically between 40 and 60 percent.
Welding is the biggest challenge with these alloys. The ferrite phase has a much greater tendency to form the brittle intermetallic phases, such as sigma, during exposure to intermediate temperatures than does austenite. Sigma causes room temperature embrittlement, and when present in appreciable amounts, lowers corrosion resistance.
Because duplex SS is sensitive to cooling rates, it is also more sensitive to welding procedures, particularly heat input. Welding procedures and heat treatments must be taken into consideration to obtain sound, corrosion-resistant welds.
Nickel alloys often are specified for many aggressive-corrosion environments. There are many classes of nickel alloys, and each has its own unique corrosion-resistant properties and welding characteristics. As with SS, improper heat treatments and welding can corrode these alloys in certain environments.
It is interesting to note that nickel alloys do not undergo a phase transformation, such as martensite formation, when cooled from high temperatures. Normally they do not need to be preheated or postweld heat-treated since they do not rely on carbon-related phases to achieve high strength at low temperatures. A postweld heat treatment usually is not necessary. Cooling rates are not critical, and a normal air cool usually is adequate for most of the nickel alloys.
The structure of a weldment is similar to that of a casting. The structure may have a coarse dendritic structure, with chemical inhomogeneity, that can cause the weld metal to become anodic to the base metal in corrosive environments.
Thus, a relatively small anode (weld metal) and a large cathode (base metal) can lead to an unfavorable galvanic corrosion situation. The weld metal characteristic also can lead to preferential general corrosion or pitting and crevice corrosion of the weld metal in environments in which the corrosion resistance of an alloy is being pushed to its limit.
One way to prevent preferential attack of as-welded structures is to use an overmatching filler wire, for example, using INCO-Weld 686CPT for welding Ni-Cr-Mo alloys C-276 or C-22. The high alloy levels (Cr + Mo + W) in 686CPT results in increased resistance to pitting, crevice, and general corrosion.
Fabricators new to the nickel alloys are encouraged to contact producers or metallurgical consultants before undertaking complex fabrications. Most nickel-alloy mills have a wealth of information on the successful fabrication and use of these alloys.
Most components used in corrosive environments are fabricated by welding. The fabricator's role in making reliable components that meet the design requirements is key to successfully using corrosion-resistant alloys and thus reducing the cost of corrosion.
The key to a successful fabrication is to understand the impact of the design, welding techniques, and metallurgy on the mechanical and corrosion properties of a component.
Dr. Hira Ahluwalia is president of Material Selection Resources Inc. He holds a Ph.D. in materials and corrosion engineering from Newcastle University, U.K. His areas of expertise include material selection, corrosion testing, metallurgy, and failure analysis. A 15-year ASM and NACE member, Ahluwalia is a program coordinator of the NACE Technical Coordination Committee and is a member of the Annual Conference Program Committee. He can be contacted at 8 Hester Court, Pennington, NJ 08534, 609-737-8226, hira@doctormetals.com, www.doctormetals.com.
Note
1. The FHWA report "Corrosion Costs and Preventive Strategies in the United States" details the results of a study funded under the Transportation Equity Act for the 21st Century (TEA-21), passed by the U.S. legislature in 1998. Conducted by CC Technologies Inc. with support from NACE International—The Corrosion Society, the study's objective was to develop an estimate of the total economic impact of metallic corrosion and identify national strategies to minimize it.
NACE International—The Corrosion Society is a professional technical society dedicated to reducing the impact of corrosion. With 15,000 members worldwide, NACE develops consensus industry standards, offers education and training, and offers 10 certification programs. www.nace.org.
Other organizations that provide useful information on corrosion and fabrication topics are ASM International, www.asminternational.org; the Nickel Development Institute, www.nidi.org; and Materials Technology Institute, www.mti-link.org.
 |
Corrosion Accidents
Excerpts reprinted from www.corrosion-doctors.com with permission from the Corrosion Doctors, Kingston, Ont., Canada.
Swimming Pool Roof Collapse
The unexpected collapse May 9, 1985, of a ceiling above a swimming pool in Uster, Switzerland, showed how a simple structural concept could be sensitive to the loss, through corrosion, of support from one of many hangers. Twelve people died.
The collapse was the result of chloride-induced stress corrosion cracking. The steel rods had been pitted, causing the roof to cave in. The roof collapsed in a zipperlike fashion, starting with the corroded rods. Chloride can overcome the passivity of the natural oxide film on steel's surface and is a major factor in the corrosion of reinforced concrete.
Sinking of the Erika
A large oil spill off France's Brittany coast occurred Dec. 12, 1999, when the tanker Erika broke up in gale force winds and rough seas. When the Erika broke up, it was carrying an estimated 30,000 tons of heavy fuel oil, 10,000 tons of which were spilled. This was equal to the total amount of oil spilled worldwide in 1998.
Corrosion problems had been apparent on the Erika since at least 1994. Severe corrosion had been discovered just weeks before the incident; however, no immediate remedial action had been taken.
Sewer Explosion due to Corrosion
A sewer explosion killed 215 people in Guadalajara, Mexico, in April 1992. Besides the fatalities, the series of blasts damaged 1,600 buildings and injured 1,500 people. Damage costs were estimated at $75 million.
At least nine separate explosions were said to have ripped a jagged trench almost 2 kilometers long, contiguous with the city sewer system. A bus was swallowed up by a hole 50 meters in diameter.
The sewer explosion was traced to the installation of a water pipe that leaked water on a gasoline line lying underneath. The subsequent corrosion of the gasoline pipeline, in turn, caused leakage of gasoline into the sewers.
About the Author
subscribe now

The Fabricator is North America's leading magazine for the metal forming and fabricating industry. The magazine delivers the news, technical articles, and case histories that enable fabricators to do their jobs more efficiently. The Fabricator has served the industry since 1970.
start your free subscription- Stay connected from anywhere

Easily access valuable industry resources now with full access to the digital edition of The Fabricator.

Easily access valuable industry resources now with full access to the digital edition of The Welder.

Easily access valuable industry resources now with full access to the digital edition of The Tube and Pipe Journal.
- Podcasting
- Podcast:
- The Fabricator Podcast
- Published:
- 04/16/2024
- Running Time:
- 63:29
In this episode of The Fabricator Podcast, Caleb Chamberlain, co-founder and CEO of OSH Cut, discusses his company’s...
- Industry Events
16th Annual Safety Conference
- April 30 - May 1, 2024
- Elgin,
Pipe and Tube Conference
- May 21 - 22, 2024
- Omaha, NE
World-Class Roll Forming Workshop
- June 5 - 6, 2024
- Louisville, KY
Advanced Laser Application Workshop
- June 25 - 27, 2024
- Novi, MI































