Contributing Writer
- FMA
- The Fabricator
- FABTECH
- Canadian Metalworking
Categories
- Additive Manufacturing
- Aluminum Welding
- Arc Welding
- Assembly and Joining
- Automation and Robotics
- Bending and Forming
- Consumables
- Cutting and Weld Prep
- Electric Vehicles
- En Español
- Finishing
- Hydroforming
- Laser Cutting
- Laser Welding
- Machining
- Manufacturing Software
- Materials Handling
- Metals/Materials
- Oxyfuel Cutting
- Plasma Cutting
- Power Tools
- Punching and Other Holemaking
- Roll Forming
- Safety
- Sawing
- Shearing
- Shop Management
- Testing and Measuring
- Tube and Pipe Fabrication
- Tube and Pipe Production
- Waterjet Cutting
Industry Directory
Webcasts
Podcasts
FAB 40
Advertise
Subscribe
Account Login
Search
Metallurgy Matters: The structure of metal
- By Bob Capudean
- Updated January 25, 2023
- April 24, 2003
- Article
- Metals/Materials

As a molten metal cools, atomic forces begin to pull or force the atoms into solid particles called nuclei, which take on specific and identifiable crystal structures.
As you think about molten metal, keep a couple of points in mind. First, heat flows to cold-always. And that becomes more understandable when you consider that warm atoms are moving faster than cold atoms. And those fast-moving atoms are bumping into other atoms, causing them to move quickly.
Furthermore, the warmer a metal-or any material, for that matter-is, the faster the atoms composing that metal are moving. Yes, there are internal attractions that help keep the atoms in a puddle, preventing them from just vaporizing, but the fact is, if they get moving fast enough-that is, get hot enough-they eventually will evaporate, just like hydrogen and oxygen do when water boils.
As thermal energy is transferred to another part, the atoms give up energy, slowing down and cooling. What evaporates is still water, in the form of steam.
As a molten metal cools, atomic forces begin to pull or force the atoms into solid particles called nuclei, which take on specific and identifiable crystal structures. Because the nuclei have the metal's crystal structure, additional atoms join the nuclei. As these nuclei get bigger, they form grains. This orderly arrangement of the atoms is called a lattice.
But as the metal solidifies and the grains grow, they grow independently of each other, which means eventually these different areas of growing grains have to meet. When they do, the arrangement of the atoms in the grain structure is disrupted at that meeting point. This is called a grain boundary. Grain boundaries form a continuous network throughout the metal, and because of the disrupted structure at the boundary, the metal often acts differently at the boundary locations.
Grain boundaries aside, each grain in a pure metal has the same crystalline structure as any other grain, assuming the temperature is the same. This structure, which is identifiable under the microscope, has a huge influence on the metal's characteristics.
Common Metal Crystal Structures
For our purposes, all metals and alloys are crystalline solids, although some metals have been formed in the lab without crystalline structure. And most metals assume one of three different lattice, or crystalline, structures as they form: body-centered cubic (BCC), face-centered cubic (FCC), or hexagonal close-packed (HCP). The atomic arrangement for each of these structures is shown in Figure 1.
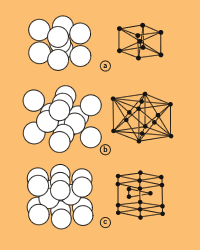 |
| Figure 1 Three crystal structures favored by metals are (a) body-centered cubic (BCC), (b) face-centered cubic (FCC), and (c) hexagonal close-packed (HCP). |
A number of metals are shown below with their room temperature crystal structure indicated. And for the record, yes, there are substances without crystalline structure at room temperature; for example, glass and silicone.
- Aluminum — FCC
- Chromium — BCC
- Copper — FCC
- Iron (alpha) — FCC
- Iron (gamma) — BCC
- Iron (delta) — BCC
- Lead — FCC
- Nickel — FCC
- Silver — FCC
- Titanium — HCP
- Tungsten — BCC
- Zinc — HCP
Alloys and Atomic Arrangements in the Structure of Metal
Everything covered so far applies to pure metals, which begs the question, What happens when you add an alloy or two? After all, most common metals are alloys containing residual and added metallic and nonmetallic elements dissolved in a base metal.
Of course, those added elements can have a dramatic effect on the resulting alloy's properties. But how those elements dissolve, or in other words how they combine with the existing atoms in the parent metal's crystal lattice, can also greatly influence both the physical and nonphysical properties of the end product.
Basically, there are two ways the alloying element(s)-called solutes-combine with the base, or parent, metal, which is also called the solvent. The alloy's atoms can combine through either direct substitution, creating a substitutional solid solution, or they can combine interstitially, forming an interstitial solid solution.
Substitutional Solid Solution. When the alloy's atoms are similar to the parent metal's atoms, they'll simply replace some of the parent metal's atoms in the lattice. The new metal dissolves in the base metal to form a solid solution. Examples include copper dissolved in nickel, gold dissolved in silver, and carbon dissolved in iron (ferrite).
Interstitial Solid Solution. When the alloy's atoms are smaller than the parent metal's atoms, they'll fit between the atoms in the parent metal's lattice. The alloy atoms don't occupy lattice sites and don't replace any of the original atoms. Of course, this causes strain in the crystal structure because the fit isn't perfect: There are atoms taking up space that was originally unoccupied.
The end result is usually an increase in tensile strength and a decrease in elongation. Examples include small amounts of copper dissolved in aluminum and carbon, and nitrogen dissolved in iron and other metals.
Phases, Microstructures, and Phase Changes
Often neither direct nor interstitial solution can completely dissolve all the added atoms. And when this happens, the result is mixed atomic groupings. In other words, different crystalline structures exist within the same alloy. Each of these different structures is called a phase, and the alloy-which is a mixture of these different crystalline structures-is called a multiphase alloy.
These different phases can be distinguished under a microscope when the alloy is polished and etched. Pearlite is a good example of a multiphase alloy within the carbon-iron family.
The phases present in an alloy, along with the overall grain arrangements and grain boundaries, combine to make up an alloy's microstructure. And the microstructure of an alloy is critical, being largely responsible for both the physical and mechanical properties of that alloy.
For example, because the boundary areas are the last to freeze when an alloy cools, grain boundaries contain lower-melting-point atoms compared to the atoms within the grains. These foreign atoms cause microstructure distortion and harden the alloy at room temperature. But as temperature goes up, alloy strength goes down because these lower-melting-point atoms begin to melt sooner, allowing slippage between the grains.
Furthermore, foreign or odd-sized atoms tend to congregate at grain boundaries because the atomic structure is irregular. This can lead to phases that reduce ductility and lead to cracking during welding.
Consider this: Cold working a metal distorts its entire microstructure. The end result, in most cases, is that the metal gets harder. Atoms from an alloying element distort the metal's microstructure, and again, the metal gets harder. The same is true for alloy atoms that are dissolved in a base metal and then precipitate out. The atoms leave, but a distortion remains, and the metal is harder.
Grain size is also important. Generally speaking, fine-grained metals have better properties at room temperature. And size is determined by cooling rate. Fast cooling leads to smaller grains, and vice versa. But the fact is, grain size, grain boundary structure, and phases present all are important. Overall, these characteristics in total determine a metal's capabilities and usefulness.
In short, a metal's overall microstructure determines its characteristics. Today just about every metal we use is an alloy, with one or more elements added to modify, adjust, correct, or change the base metal's microstructure, creating a multiphase system that can better serve our needs. And every time we put torch to metal, we cause a phase change and influence that microstructure.
This should give you an overview of how metals are structured and what happens when we melt them to weld them together. Next time we'll consider phase transformations, carbon content, hardening, the relationship between austenite and martensite, and the influence of welding on metallurgical structure.
About the Author

Bob Capudean
Back Alley Customs
About the Publication
subscribe now

The Welder, formerly known as Practical Welding Today, is a showcase of the real people who make the products we use and work with every day. This magazine has served the welding community in North America well for more than 20 years.
start your free subscription- Stay connected from anywhere

Easily access valuable industry resources now with full access to the digital edition of The Fabricator.

Easily access valuable industry resources now with full access to the digital edition of The Welder.

Easily access valuable industry resources now with full access to the digital edition of The Tube and Pipe Journal.
- Podcasting
- Podcast:
- The Fabricator Podcast
- Published:
- 04/16/2024
- Running Time:
- 63:29
In this episode of The Fabricator Podcast, Caleb Chamberlain, co-founder and CEO of OSH Cut, discusses his company’s...
- Trending Articles
Sheffield Forgemasters makes global leap in welding technology

ESAB unveils Texas facility renovation

Engine-driven welding machines include integrated air compressors

How welders can stay safe during grinding

The impact of sine and square waves in aluminum AC welding, Part I

- Industry Events
16th Annual Safety Conference
- April 30 - May 1, 2024
- Elgin,
Pipe and Tube Conference
- May 21 - 22, 2024
- Omaha, NE
World-Class Roll Forming Workshop
- June 5 - 6, 2024
- Louisville, KY
Advanced Laser Application Workshop
- June 25 - 27, 2024
- Novi, MI