Tube and Pipe Product Manager
- FMA
- The Fabricator
- FABTECH
- Canadian Metalworking
Categories
- Additive Manufacturing
- Aluminum Welding
- Arc Welding
- Assembly and Joining
- Automation and Robotics
- Bending and Forming
- Consumables
- Cutting and Weld Prep
- Electric Vehicles
- En Español
- Finishing
- Hydroforming
- Laser Cutting
- Laser Welding
- Machining
- Manufacturing Software
- Materials Handling
- Metals/Materials
- Oxyfuel Cutting
- Plasma Cutting
- Power Tools
- Punching and Other Holemaking
- Roll Forming
- Safety
- Sawing
- Shearing
- Shop Management
- Testing and Measuring
- Tube and Pipe Fabrication
- Tube and Pipe Production
- Waterjet Cutting
Industry Directory
Webcasts
Podcasts
FAB 40
Advertise
Subscribe
Account Login
Search
Understanding coating adhesion
How metalworking fluids, corrosion preventives interact
- By Mark Diaz and William R. Skowronek
- August 15, 2002
- Article
- Tube and Pipe Fabrication

Figure 1: Pipes typically are bundled and moved outside for storage, regardless of weather, often within an hour of being coated. Rapid curing is essential to ensure that the pipes resist corrosion and withstand handling by the equipment used to move them.
Producers of tubular products used in oil field production, construction, and manufactured goods are responsible for manufacturing, storing, and shipping these materials to the end user free of rust and corrosion. This can be accomplished using a variety of corrosion preventive coatings, depending on the tube's final use and storage conditions. With today's concerns about costs, worker safety, and environmental restrictions, choosing the right coating for a particular tube and pipe application can pose a challenge.
Studying the Market
In 1999 Quaker Chemical Corp. conducted a market study to uncover the current and future needs of worldwide tube and pipe producers regarding corrosion preventive coating application and performance. As a result of this study, the company was able to segregate coating requirements into two distinct tubular product lines: seamless tube and pipe and electrical resistance-welded (ERW) tube and pipe.
Seamless Tube and Pipe. Because most seamless tube and pipe is large in diameter, outdoor storage is a common practice. The coatings used for seamless products are predominantly colorless, because stencil markings on the tube must be visible for the product's entire life. Coatings are considered permanent and must be able to withstand corrosion despite outdoor storage (see Figure 1).
ERW Tube. This type of tube is used in applications such as furniture, fuel line, decorative, and general mechanical. The specific use influences the type of corrosion protection needed. In some cases, the coatings may need to be pigmented, which adds another level of complexity. Coatings for ERW tubing can be classed three ways:
- They can serve as a primer. Many end users, such as furniture manufacturers, use the tube as is and add a decorative finish. Therefore, it's important to ensure that a variety of topcoats will adhere to the final mill coating.
- They can provide long-term corrosion protection in harsh outdoor conditions. If no additional coatings will be applied, then the final mill coating must perform similarly to that used on seamless pipe.
- They can provide interim corrosion protection. If additional coatings will be applied, the coating must offer temporary protection until the final finishing process takes place. Generally, tube or pipe that has an interim corrosion protection is stored under better conditions. The coating must be easily removed with a mild alkaline cleaning solution.
More About Coatings
The market study revealed that alkyd- and acrylic-based coating resins cover the majority of tubular goods. Depending on regional regulations, either water- or solventborne coating formulations are used, with an ever-growing trend toward using waterborne coatings as the industry works to comply with environmental regulations.
To understand the implications this trend has on coating performance, it's necessary to have a more comprehensive understanding of metalworking fluids used in tube and pipe production and how they interact with waterborne coating systems. Some basic questions and answers can serve to develop this understanding.
How Does a Coating Provide Corrosion Protection? A fully cured coating protects a tube or pipe by forming a barrier that water or water vapor cannot penetrate easily. This barrier of protection helps to seal the steel surface and provides an additional layer of protection between the steel and the elements. While the coating itself is not totally impermeable by the elements, proper product selection can maximize film performance.
During the application process or subsequent handling of the tube, the coating can develop surface defects as varied as entrapped air bubbles and scratches to roller marks and chips. These defects then can provide a pathway for water or water vapor to reach the surface and begin the corrosion process. The critical link between surface defects and corrosion protection is adhesion.
Adhesion, which refers to the coating's bond to the metal surface, determines how fast, and to what extent, oxidation will spread across the steel surface. The more tenacious the coating's adhesion, the more difficult it is for corrosion to spread. A coating with poor adhesion rapidly delaminates from the surface, allowing oxidation to spread quickly.
What Influences a Coating's Adhesion? Coatings applied to tubular products form a mechanical bond with the steel surface. The tube's texture and cleanness are the key elements that can affect adhesion. In general, the rougher the metal surface, the greater the adhesion.
However, imperfections on the metal surface, which may make it appear rougher and therefore more desirable for adhesion, can be detrimental. These imperfections may delaminate from the metal surface after coating and provide a passageway for corrosion to begin.
Surface cleanness also is critical to coating adhesion but often is overlooked in the coating process. Not only can surface contaminants affect the aesthetic appearance of the coating, but they can lead to poor film adhesion as well. Surface contaminants can create different surface tensions in the film, leading to film voids. These voids, commonly called fish eyes, cause rapid corrosion formation.
Lack of cleanness may not be detectable during coating application, but it becomes evident when a coating does not adhere during a production run.
The type of coating system used also plays a role. Traditional hydrocarbon solvents tend to dissolve many forms of surface contamination, encouraging better adhesion. Because waterborne coatings are not as forgiving, surface cleanness is even more critical with this type of coating.
Sources of Surface Contamination
A variety of metalworking fluids are used during tube production. They are:
- Corrosion preventive oils that protect the steel used in ERW mills.
- Coolants used in ERW mills.
- Coolants used with cutoff shears and saws.
- Cleaners and interim corrosion preventive coatings used during the manufacturing process.
- Ink and solvent residue from the stenciling process.
- Lubricants that leak from mills.
These fluids, which are neat oil or water-soluble, can be used in several processes before final coating. Once applied, they generally are not removed. The resulting residues, which consist largely of mineral oil, are the primary sources of contaminants in the production process and can have a drastic effect on coating adhesion, particularly in waterborne systems.
How Metalworking Fluids Influence Coating Adhesion and Corrosion Resistance
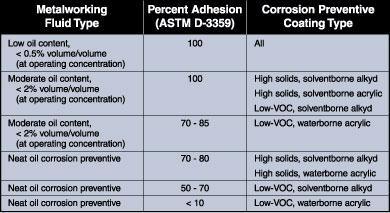
To understand better the interactions, the market study team examined several types of metalworking fluids and corrosion preventive fluids (see Figure 2). Then the team developed a procedure for quantifying the effects of adhesion and corrosion protection on pipe coatings.
ERW and seamless pipes were cut into 30-centimeter lengths and thoroughly cleansed of surface contaminants and scale. The pipes then were dipped into the various metalworking fluids, suspended for an hour to allow excess fluid to run off, and allowed to dry at ambient temperature for 24 hours.
A candidate coating then was applied to each test piece via immersion. Unlike atomization spray techniques such as airless or air spray, immersion application highlights surface contaminant-induced defects. The coated pipes then were suspended for an hour to allow excess coating to run off and allowed to dry at ambient temperature for seven days.
At the end of that period, a tape adhesive method established by the American Society for Testing and Materials (ASTM, www.astm.org) was used to test the adhesion performance of the various combinations of coolants and coatings.
The study showed that the coating adhesion was most affected by the amount of oil present in the metalworking fluid. All coating types tested had 100 percent adhesion when paired with a metalworking fluid low in oil content. The lowest percentage of coating adhesion (less than 10 percent) occurred when neat oil corrosion preventive fluids were paired with low-VOC, waterborne acrylic coatings.
The Growing Prevalence of Waterborne Coatings
As environmental regulations restricting solvent emissions into the atmosphere become stricter, tube and pipe producers will be moving toward waterborne coatings to ensure compliance. As this study demonstrates, metalworking fluids have a significant impact on the adhesion of waterborne coatings.
A better understanding of metalworking fluid and coating interactions should help the industry better adapt to changing technologies and environmental regulations.
About the Authors
Mark Diaz
One Quaker Park 901 Hector St.
Conshohocken, PA 19428
610-832-4000
William R. Skowronek
Metalworking Marketing Manager
One Quaker Part 901 Hector St.
Conshohocken, PA 19428
610-832-4000
About the Publication
Related Companies
subscribe now

The Tube and Pipe Journal became the first magazine dedicated to serving the metal tube and pipe industry in 1990. Today, it remains the only North American publication devoted to this industry, and it has become the most trusted source of information for tube and pipe professionals.
start your free subscription- Stay connected from anywhere

Easily access valuable industry resources now with full access to the digital edition of The Fabricator.

Easily access valuable industry resources now with full access to the digital edition of The Welder.

Easily access valuable industry resources now with full access to the digital edition of The Tube and Pipe Journal.
- Podcasting
- Podcast:
- The Fabricator Podcast
- Published:
- 04/16/2024
- Running Time:
- 63:29
In this episode of The Fabricator Podcast, Caleb Chamberlain, co-founder and CEO of OSH Cut, discusses his company’s...
- Trending Articles
Zekelman Industries to invest $120 million in Arkansas expansion

3D laser tube cutting system available in 3, 4, or 5 kW

Corrosion-inhibiting coating can be peeled off after use

Brushless copper tubing cutter adjusts to ODs up to 2-1/8 in.

HGG Profiling Equipment names area sales manager

- Industry Events
16th Annual Safety Conference
- April 30 - May 1, 2024
- Elgin,
Pipe and Tube Conference
- May 21 - 22, 2024
- Omaha, NE
World-Class Roll Forming Workshop
- June 5 - 6, 2024
- Louisville, KY
Advanced Laser Application Workshop
- June 25 - 27, 2024
- Novi, MI